Recent Works
- “Quantum Chemical Calculations to Trace Back Reaction Paths for the Prediction of Reactants”, Sumiya, Y.; Harabuchi, Y.; Nagata, Y.; Maeda, S. JACS Au 2022, 2, 1181-1188. 10.1021/jacsau.2c00157
- “Tyrosine-based photoluminescent diketopiperazine supramolecular aggregates”, Shimosaraya, N.; Sotani, T.; Miyagi, Y.; Mondarte, E. A. Q.; Suthiwanich, K.; Hayashi, T.; Nagata, Y.; Sogawa, H.; Sanda, F. Soft Matter 2022, 18, 137-145. 10.1039/D1SM01206A
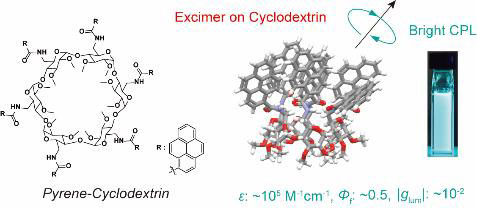
- “Cyclodextrins with Multiple Pyrenyl Groups: An Approach to Organic Molecules Exhibiting Bright Excimer Circularly Polarized Luminescence”, Shigemitsu, H.; Kawakami, K.; Nagata, Y.; Kajiwara, R.; Yamada, S.; Mori, T.; Kida, T. Angew. Chem. Int. Ed. 2022, 61, e202114700. 10.1002/anie.202114700
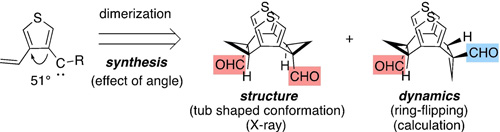
- “Synthesis, Structure, and Dynamics of Chiral Eight-Membered Cyclic Molecules with Thienylene and Cyclopropylene Units Alternately Connected”, Miura, T.; Ishihara, Y.; Nakamuro, T.; Moritani, S.; Nagata, Y.; Murakami, M. Chemistry 2022, 28, e202103962. 10.1002/chem.202103962
- “Preparative-scale synthesis of nonacene”, Jancarik, A.; Holec, J.; Nagata, Y.; Samal, M.; Gourdon, A. Nat. Commun. 2022, 13, 223. 10.1038/s41467-021-27809-0
- “Alkali Metal Fluorides in Fluorinated Alcohols: Fundamental Properties and Applications to Electrochemical Fluorination”, Shida, N.; Takenaka, H.; Gotou, A.; Isogai, T.; Yamauchi, A.; Kishikawa, Y.; Nagata, Y.; Tomita, I.; Fuchigami, T.; Inagi, S. J. Org. Chem. 2021, 86, 16128-16133. 10.1021/acs.joc.1c00692
- “Stereo- and Enantioselective Synthesis of Propionate-Derived Trisubstituted Alkene Motifs”, Miura, T.; Oku, N.; Shiratori, Y.; Nagata, Y.; Murakami, M. Chemistry 2021, 27, 3861-3868. 10.1002/chem.202004930

- “Synthesis and Absorption Properties of Long Acenoacenes”, Jancarik, A.; Mildner, D.; Nagata, Y.; Banasiewicz, M.; Olas, J.; Kozankiewicz, B.; Holec, J.; Gourdon, A. Chem. Eur. J. 2021, 27, 12388-12394. 10.1002/chem.202101577

- “Protected amino acids as a nonbonding source of chirality in induction of single-handed screw-sense to helical macromolecular catalysts”, Ikeda, S.; Takeda, R.; Fujie, T.; Ariki, N.; Nagata, Y.; Suginome, M. Chem. Sci. 2021,12, 8811-8816. 10.1039/d1sc01764k
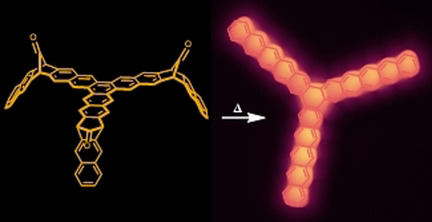
- “A Large Starphene Comprising Pentacene Branches”, Holec, J.; Cogliati, B.; Lawrence, J.; Berdonces-Layunta, A.; Herrero, P.; Nagata, Y.; Banasiewicz, M.; Kozankiewicz, B.; Corso, M.; de Oteyza, D. G.; Jancarik, A.; Gourdon, A. Angew. Chem., Int. Ed. 2021, 60, 7752-7758. 10.1002/anie.202016163
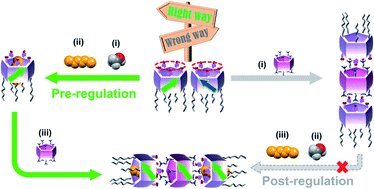
- “Pre-regulation of the planar chirality of pillar[5]arenes for preparing discrete chiral nanotubes”, Fa, S. X.; Adachi, K.; Nagata, Y.; Egami, K.; Kato, K.; Ogoshi, T. Chem. Sci. 2021, 12, 3483-3488. 10.1039/d1sc00074h
- “Stereo- and Enantioselective Synthesis of Propionate-Derived Trisubstituted Alkene Motifs”, Miura, T.; Oku, N.; Shiratori, Y.; Nagata, Y.; Murakami, M. Chem. Eur. J. 2021, 27, 3861-3868. 10.1002/chem.202004930

- “Regioselective 1,3-Dipolar Cycloaddition of Nitriles with Nitrile Imines Generated from Tetrazoles”, Miura, T.; Hagiwara, K.; Nakamuro, T.; Nagata, Y.; Oku, N.; Murakami, M. Chem. Lett. 2020. 10.1246/cl.200634

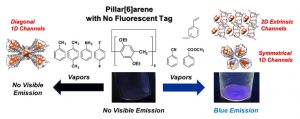
- “Vapoluminescence Behavior Triggered by Crystal State Complexation between Host Crystals and Guest Vapors Exhibiting No Visible Fluorescence”, Ogoshi, T.; Hamada, Y.; Sueto, R.; Kojima, R.; Sakakibara, F.; Nagata, Y.; Sakata, Y.; Akine, S.; Ono, T.; Kakuta, T.; Yamagishi, T. Cryst. Growth Des. 2020, 20, 7087-7092. 10.1021/acs.cgd.0c00982
- “Chiral Macrocycles Having C3 Symmetry Owing to Orientation of Thiophenes”, Miura, T.; Nakamuro T.; Ishihara, Y.; Nagata, Y.; Murakami, M. Angew. Chem. Int. Ed. 2020, 59, 20475 –20479. 10.1002/anie.202009781
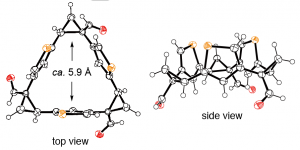
- “Cubane Chirality via Substitution of a “Hidden” Regular Tetrahedron”, Yoshino, N.; Kato, Y.; Mabit, T.; Nagata, Y.; Williams, C. M.; Harada, M.; Muranaka, A.; Uchiyama, M.; Matsubara, S. Org. Lett. 2020, 22, 4083-4087. 10.1021/acs.orglett.0c01142
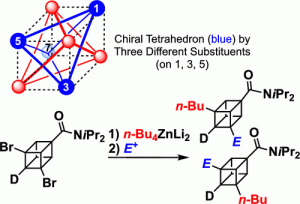
- “Holding of planar chirality of pillar[5]arene by kinetic trapping using host–guest interactions with achiral guest solvents”, Nagata, Y.; Suzuki, M.; Shimada, Y.; Sengoku, H.; Nishida, S.; Kakuta, T.; Yamagishi, T.; Suginome, M.; Ogoshi, T. Chem. Commun. 2020, 56, 8424-8427. 10.1039/d0cc03413d

- “Irreverent Nature of Dissymmetry Factor and Quantum Yield in Circularly Polarized Luminescence of Small Organic Molecules”, Nagata, Y.; Mori, T. Frontiers in Chemistry 2020, 8, 448. 10.3389/fchem.2020.00448

Previous Works (until Dec. 2019)
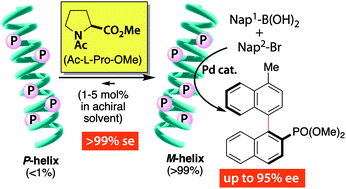
- “Asymmetric Catalysis in Chiral Solvents: Chirality Transfer with Amplification of Homochirality through a Helical Macromolecular Scaffold”, Nagata, Y.; Takeda, R.; Suginome, M. ACS Central Science 2019, 5, 1235-1240. 10.1021/acscentsci.9b00330

- “A bidirectional screw-sense induction of poly(quinoxaline-2,3-diyl)s that depends on the degree of polymerization”, Nagata, Y.; Nishikawa, T.; Terao, K.; Hasegawa, H.; Suginome, M. J. Polym. Sci., Part A: Polym. Chem. 2019, 57, 260-263. 10.1002/pola.29224

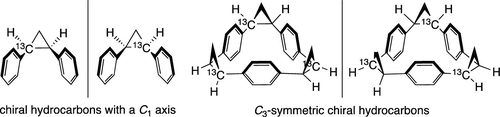
- “Asymmetric Synthesis and Stereochemical Assignment of C12/C13 Isotopomers”, Miura, T.; Nakamuro, T.; Nagata, Y.; Moriyama, D.; Stewart, S. G.; Murakami, M. J. Am. Chem. Soc. 2019, 141, 13341-13345. 10.1021/jacs.9b07181
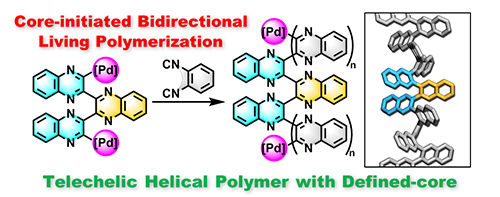
- “Telechelic Helical Poly(quinoxaline-2,3-diyl)s Containing a Structurally Defined, Circularly Polarized Luminescent Terquinoxaline Core: Synthesis by Core-Initiated Bidirectional Living Polymerization”, Kuriyama, S.; Nagata, Y.; Suginome, M. ACS Macro Lett. 2019, 8, 479-485. 10.1021/acsmacrolett.9b00165
- “Control of Circularly Polarized Luminescence by Orientation of Stacked p-Electron Systems”, Kikuchi, K.; Nakamura, J.; Nagata, Y.; Tsuchida, H.; Kakuta, T.; Ogoshi, T.; Morisaki, Y. Chem-Asian J 2019, 14, 1681-1685. 10.1002/asia.201801741

- “Lyotropic Liquid Crystallinity of Linear and Star Poly(quinoxaline-2,3-diyl)s: Isotropic-Liquid Crystal Phase Equilibria in Tetrahydrofuran”, Hasegawa, H.; Terao, K.; Sato, T.; Nagata, Y.; Suginome, M. Macromolecules 2019, 52, 3158-3164. 10.1021/acs.macromol.9b00460

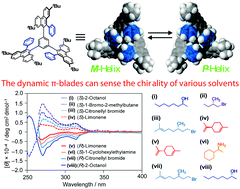
- “Sensing the chirality of various organic solvents by helically arranged p-blades”, Shimizu, Y.; Shoji, Y.; Hashizume, D.; Nagata, Y.; Fukushima, T. Chem. Commun. 2018, 54, 12314-12317. 10.1039/c8cc06277c
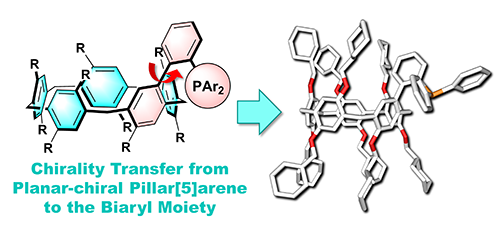
- “A Planar-Chiral Pillar[5]arene-Based Monophosphine Ligand with Induced Chirality at the Biaryl Axis”, Nagata, Y.; Shimada, Y.; Nishikawa, T.; Takeda, R.; Uno, M.; Ogoshi, T.; Suginome, M. Synlett 2018, 29, 2167-2170. 10.1055/s-0037-1610635
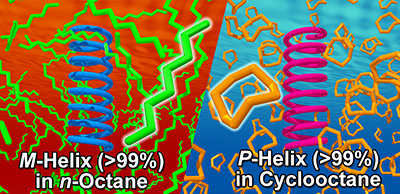
- “Elucidating the Solvent Effect on the Switch of the Helicity of Poly(quinoxaline-2,3-diyl)s: A Conformational Analysis by Small-Angle Neutron Scattering”, Nagata, Y.; Nishikawa, T.; Suginome, M.; Sato, S.; Sugiyama, M.; Porcar, L.; Martel, A.; Inoue, R.; Sato, N. J. Am. Chem. Soc. 2018, 140, 2722-2726. 10.1021/jacs.7b11626

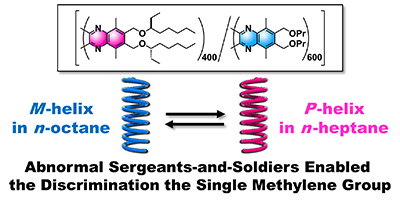
- “Abnormal sergeants-and-soldiers effects of poly(quinoxaline-2,3-diyl)s enabling discrimination of one-carbon homologous n-alkanes through a highly sensitive solvent-dependent helix inversion”, Nagata, Y.; Nishikawa, T.; Suginome, M. Chem. Commun. 2018, 54, 6867-6870. 10.1039/c8cc02836b
- “Poly(quinoxaline-2,3-diyl) as a Multifunctional Chiral Scaffold for Circularly Polarized Luminescent Materials: Color Tuning, Energy Transfer, and Switching of the CPL Handedness”, Nishikawa, T.; Nagata, Y.; Suginome, M. ACS Macro Lett. 2017, 6, 431-435. 10.1021/acsmacrolett.7b00131

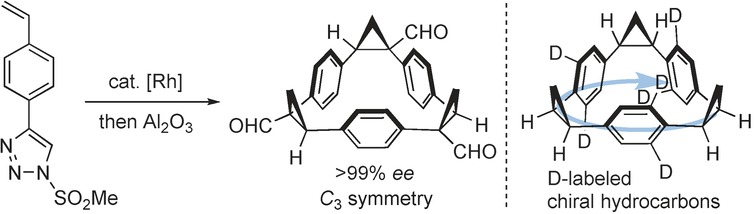
- “Synthesis of Enantiopure C-3-Symmetric Triangular Molecules”, Miura, T.; Nakamuro, T.; Stewart, S. G.; Nagata, Y.; Murakami, M. Angew. Chem. Int. Ed. 2017, 56, 3334-3338. 10.1002/anie.201612585
- “Synthesis and Catalytic Applications of a Triptycene-Based Monophosphine Ligand for Palladium-Mediated Organic Transformations”, Leung, F. K. C.; Ishiwari, F.; Shoji, Y.; Nishikawa, T.; Takeda, R.; Nagata, Y.; Suginome, M.; Uozumi, Y.; Yamada, Y. M. A.; Fukushima, T. Acs Omega 2017, 2, 1930-1937. 10.1021/acsomega.7b00200
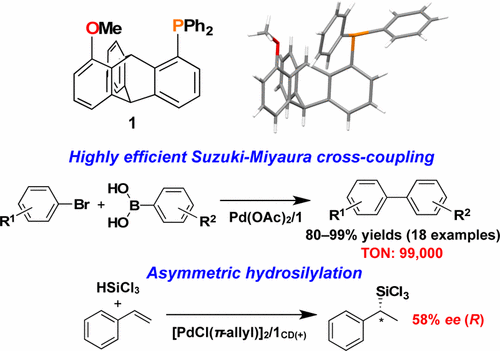
- “Synthesis and Solution Properties of a Rigid Helical Star Polymer: Three-Arm Star Poly(quinoxaline-2,3-diyl)”, Hasegawa, H.; Nagata, Y.; Terao, K.; Suginome, M. Macromolecules 2017, 50, 7491-7497. 10.1021/acs.macromol.7b01797

- “Three-Way-Switchable (Right/Left/OFF) Selective Reflection of Circularly Polarized Light on Solid Thin Films of Helical Polymer Blends”, Nagata, Y.; Uno, M.; Suginome, M. Angew. Chem. Int. Ed. 2016, 55, 7126-7130. 10.1002/anie.201602035

- “Solvent Effect on the Sergeants-and-Soldiers Effect Leading to Bidirectional Induction of Single-Handed Helical Sense of Poly(quinoxaline-2,3-diyl)s Copolymers in Aromatic Solvents”, Nagata, Y.; Nishikawa, T.; Suginome, M. ACS Macro Lett. 2016, 5, 519-522. 10.1021/acsmacrolett.6b00191
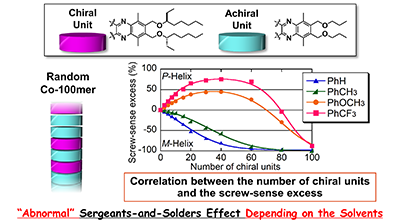
- “Pressure-dependent helix inversion of poly(quinoxaline-2,3-diyl)s containing chiral side chains in non-aqueous solvents”, Nagata, Y.; Takeda, R.; Suginome, M. Chem. Commun. 2015, 51, 11182-11185. 10.1039/c5cc04255k

- “Exerting Control over the Helical Chirality in the Main Chain of Sergeants-and-Soldiers-Type Poly(quinoxaline-2,3-diyl)s by Changing from Random to Block Copolymerization Protocols”, Nagata, Y.; Nishikawa, T.; Suginome, M. J. Am. Chem. Soc. 2015, 137, 4070-4073. 10.1021/Jacs.5b01422
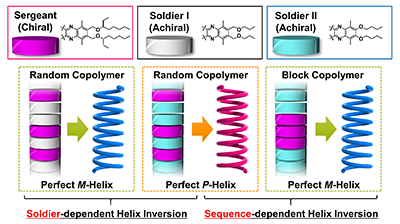
- “Facile Preparation of Poly(quinoxaline-2,3-diyl)s via Aromatizing Polymerization of 1,2-Diisocyanobenzenes Using Phosphine Complexes of Nickel(II) Salts”, Nagata, Y.; Ke, Y.-Z.; Suginome, M. Chem. Lett. 2015, 44, 53-55. 10.1246/Cl.140909

- “Main-chain stiffness and helical conformation of a poly(quinoxaline-2,3-diyl) in solution”, Nagata, Y.; Hasegawa, H.; Terao, K.; Suginome, M. Macromolecules 2015, 48, 7983-7989. 10.1021/acs.macromol.5b01919

- “Majority-Rules-Type Helical Poly(quinoxaline-2,3-diyl)s as Highly Efficient Chirality-Amplification Systems for Asymmetric Catalysis”, Ke, Y.-Z.; Nagata, Y.; Yamada, T.; Suginome, M. Angew. Chem. Int. Ed. 2015, 54, 9333-9337. 10.1002/anie.201502209
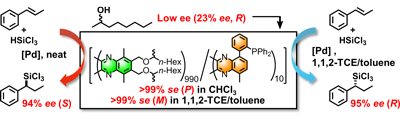
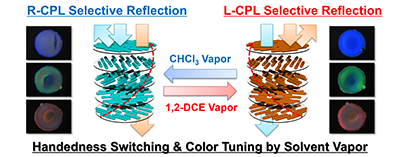
- “Solid Polymer Films Exhibiting Handedness-Switchable, Full-Color-Tunable Selective Reflection of Circularly Polarized Light”, Nagata, Y.; Takagi, K.; Suginome, M. J. Am. Chem. Soc. 2014, 136, 9858-9861. 10.1021/ja504808r
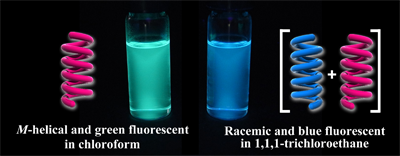
- “Chirality-Switchable Circularly Polarized Luminescence in Solution Based on the Solvent-Dependent Helix Inversion of Poly(quinoxaline-2,3-diyl)s”, Nagata, Y.; Nishikawa, T.; Suginome, M. Chem. Commun. 2014, 50, 9951-9953. 10.1039/c4cc03944k

- “Poly(quinoxaline-2,3-diyl)s bearing (S)-3-octyloxymethyl side chains as an efficient amplifier of alkane solvent effect leading to switch of main-chain helical chirality”, Nagata, Y.; Nishikawa, T.; Suginome, M. J. Am. Chem. Soc. 2014, 136, 15901-15904. 10.1021/Ja509531t
- “Ether solvent-induced chirality inversion of helical poly(quinoxaline-2,3-diyl)s containing L-lactic acid derived side chains”, Nagata, Y.; Kuroda, T.; Takagi, K.; Suginome, M. Chem. Sci. 2014, 5, 4953-4956. 10.1039/C4sc01920b

- “Solvent-Dependent Switch of Helical Main-Chain Chirality in Sergeants-and-Soldiers-Type Poly(quinoxaline-2,3-diyl)s: Effect of the Position and Structures of the “Sergeant” Chiral Units on the Screw-Sense Induction”, Nagata, Y.; Yamada, T.; Adachi, T.; Akai, Y.; Yamamoto, T.; Suginome, M. J. Am. Chem. Soc. 2013, 135, 10104-10113. 10.1021/Ja403391m

- “Control of helical chirality of poly(quinoxaline-2,3-diyl)s based on postpolymerization modification of the terminal group by small chiral molecules”, Nagata, Y.; Ohashi, S.; Suginome, M. J. Polym. Sci., Part A: Polym. Chem. 2012, 50, 1564-1571. 10.1002/Pola.25926

- “Solvent-dependent fluorescence and circular dichroism properties of poly(quinoxaline-2,3-diyl)s bearing pyrene pendants”, Nagata, Y.; Nishikawa, T.; Suginome, M. Chem. Commun. 2012, 48, 11193-11195. 10.1039/C2cc36275a
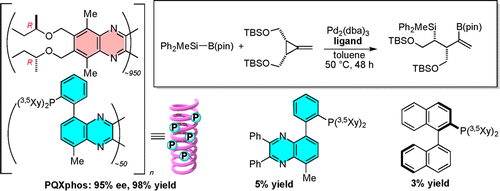
- “Enhanced Catalyst Activity and Enantioselectivity with Chirality-Switchable Polymer Ligand PQXphos in Pd-Catalyzed Asymmetric Silaborative Cleavage of meso-Methylenecyclopropanes”, Akai, Y.; Yamamoto, T.; Nagata, Y.; Ohmura, T.; Suginome, M. J. Am. Chem. Soc. 2012, 134, 11092-11095. 10.1021/Ja303506k
- “Highly Enantioselective Synthesis of Axially Chiral Biarylphosphonates: Asymmetric Suzuki-Miyaura Coupling Using High-Molecular-Weight, Helically Chiral Polyquinoxaline-Based Phosphines”, Yamamoto, T.; Akai, Y.; Nagata, Y.; Suginome, M. Angew. Chem. Int. Ed. 2011, 50, 8844-8847. 10.1002/anie.201103792

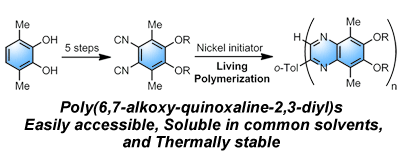
- “Synthesis of Poly(quinoxaline-2,3-diyl)s with Alkoxy Side Chains by Aromatizing Polymerization of 4,5-Dialkoxy-substituted 1,2-Diisocyanobenzenes”, Nagata, Y.; Suginome, M. J. Polym. Sci., Part A: Polym. Chem. 2011, 49, 4275-4282. 10.1002/Pola.24871
- “High-Molecular-Weight Polyquinoxaline-Based Helically Chiral Phosphine (PQXphos) as Chirality-Switchable, Reusable, and Highly Enantioselective Monodentate Ligand in Catalytic Asymmetric Hydrosilylation of Styrenes”, Yamamoto, T.; Yamada, T.; Nagata, Y.; Suginome, M. J. Am. Chem. Soc. 2010, 132, 7899-7901. 10.1021/Ja102428q

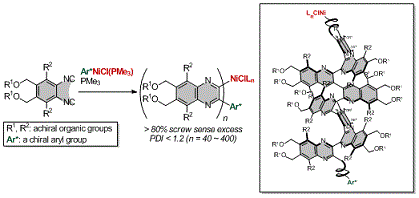
- “Chiral Arylnickel Complexes as Highly Active Initiators for Screw-Sense Selective Living Polymerization of 1,2-Diisocyanobenzenes”, Yamada, T.; Noguchi, H.; Nagata, Y.; Suginome, M. J. Polym. Sci., Part A: Polym. Chem. 2010, 48, 898-904. 10.1002/Pola.23842
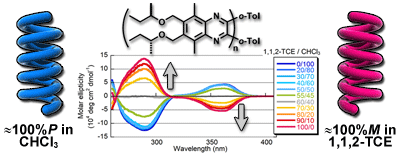
- “Non-hydrogen-bonding-based, solvent-dependent helix inversion between pure P-helix and pure M-helix in poly(quinoxaline-2,3-diyl)s bearing chiral side chains”, Yamada, T.; Nagata, Y.; Suginome, M. Chem. Commun. 2010, 46, 4914-4916. 10.1039/C001564d
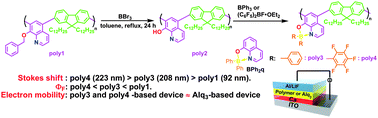
- “Luminescent alternating boron quinolate-fluorene copolymers exhibiting high electron mobility”, Nagai, A.; Kobayashi, S.; Nagata, Y.; Kokado, K.; Taka, H.; Kita, H.; Suzuri, Y.; Chujo, Y. J. Mater. Chem. 2010, 20, 5196-5201. 10.1039/B924729g
- “Synthesis and luminescent properties of pyrenylvinylene-substituted tripylborane”, Nagata, Y.; Chujo, Y. J. Organomet. Chem. 2009, 694, 1723-1726. 10.1016/j.jorganchem.2009.01.015
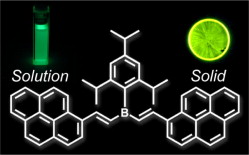
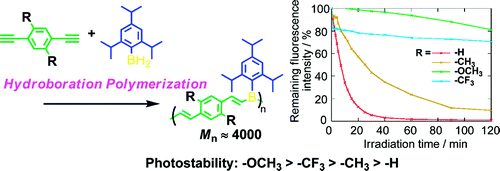
- “Synthesis and Photostability of Poly(p-phenylenevinylene-borane)s”, Nagai, A.; Murakami, T.; Nagata, Y.; Kokado, K.; Chujo, Y. Macromolecules 2009, 42, 7217-7220. 10.1021/Ma901001b
- “A Facile Synthesis of Chiral Luminescent Organoboron Polymers by Hydroboration Polymerization Utilizing Chiral Borane”, Nagai, A.; Miyake, J.; Kokado, K.; Nagata, Y.; Chujo, Y. Macromolecules 2009, 42, 1560-1564. 10.1021/Ma802257k
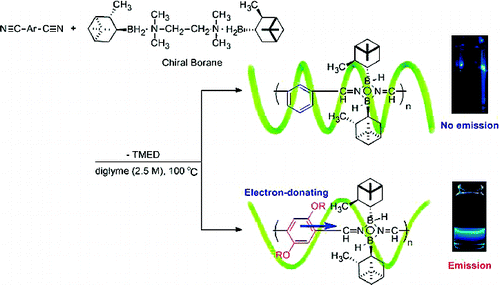
- “Synthesis of new main-chain-type organoboron quinolate polymer linked on quinolate ligand”, Nagata, Y.; Otaka, H.; Chujo, Y. Macromolecules 2008, 41, 737-740. 10.1021/Ma702346i
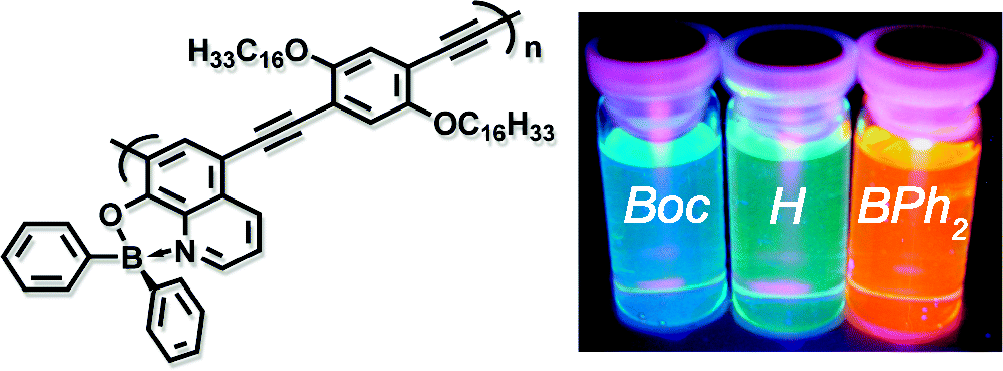
- “Main-chain-type N,N ‘-chelate organoboron ammoquinolate polymers: Synthesis, luminescence, and energy transfer behavior”, Nagata, Y.; Chujo, Y. Macromolecules 2008, 41, 3488-3492. 10.1021/Ma702873a

- “Synthesis of methyl-substituted main-chain-type organoboron quinolate, polymers and their emission color tuning”, Nagata, Y.; Chujo, Y. Macromolecules 2008, 41, 2809-2813. 10.1021/Ma7025918

- “Highly Luminescent BODIPY-Based Organoboron Polymer Exhibiting Supramolecular Self-Assemble Structure”, Nagai, A.; Miyake, J.; Kokado, K.; Nagata, Y.; Chujo, Y. J. Am. Chem. Soc. 2008, 130, 15276-15278. 10.1021/Ja806939w
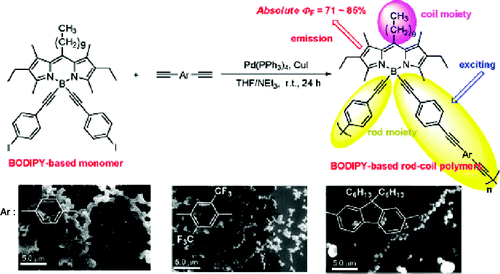
- “1,3-Diketone-Based Organoboron Polymers: Emission by Extending pi-Conjugation along a Polymeric Ligand”, Nagai, A.; Kokado, K.; Nagata, Y.; Chujo, Y. Macromolecules 2008, 41, 8295-8298. 10.1021/Ma801690d

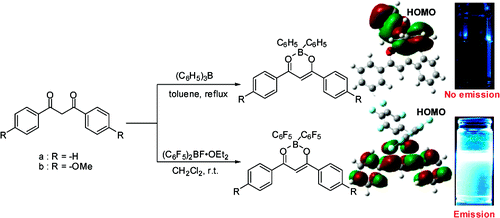
- “Highly Intense Fluorescent Diarylboron Diketonate”, Nagai, A.; Kokado, K.; Nagata, Y.; Arita, M.; Chujo, Y. J. Org. Chem. 2008, 73, 8605-8607. 10.1021/Jo8017582
- “Stereospecific construction of a trans-1,4-diphosphacyclohexane skeleton”, Morisaki, Y.; Imoto, H.; Ouchi, Y.; Nagata, Y.; Chujo, Y. Org. Lett. 2008, 10, 1489-1492. 10.1021/Ol800261j

- “Main-chain-type organoboron quinolate polymers: Synthesis and photoluminescence properties”, Nagata, Y.; Chujo, Y. Macromolecules 2007, 40, 6-8. 10.1021/Ma062007s

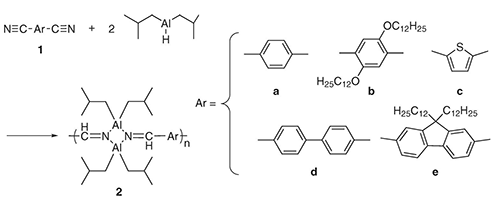
- “Synthesis of organoaluminum polymers with aluminum – nitrogen ring in their main-chain”, Nagata, Y.; Chujo, Y. Main Group Chem. 2007, 5, 287-295. 10.1080/10241220701650915
- “Synthesis of novel poly(pyrazabole)s with electron-withdrawing structure in their main chain”, Matsumoto, F.; Nagata, Y.; Chujo, Y. Polym. Bull. 2005, 53, 155-160. 10.1007/s00289-004-0327-4
